|
Therefore, while the Law of Definite Proportions can be used to compare two experiments in which hydrogen and oxygen react to form water, the Law of Definite Proportions can not be used to compare one experiment in which hydrogen and oxygen react to form water, and another experiment in which hydrogen and oxygen react to form hydrogen peroxide (peroxide is another material that can be made from hydrogen and oxygen). The Law of Definite Proportions applies when elements are reacted together to form the same product. At the beginning of the 19th century, the English scientist John Dalton proposed an atomic theory that became the basis for the study of chemistry. In 1803, Dalton published a table of atomic weights for five known elements. While his original lists of elements were limited, Dalton made significant progress in expanding our understanding of the elements during his lifetime. 
Law of Definite Proportions states that in a given type of chemical substance, the elements are always combined in the same proportions by mass. John Dalton’s contributions to the development of the periodic table extended beyond his atomic theory. Similarly, when 2 grams of A react with 16 grams of B, they must produce 18 grams of C. The law of conservation of mass says that within a closed system, no. His theory was based on two verified scientific laws: the law of conservation of mass and the law of constant composition. 
If 1 gram of A reacts with 8 grams of B, then by the Law of Conservation of Mass, they must produce 9 grams of C. John Dalton’s atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete. 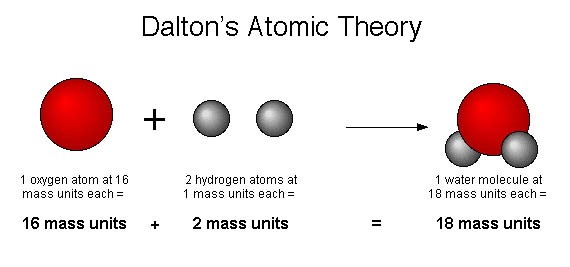
\): If 1 gram of A reacts with 8 grams of B, then by the Law of Definite Proportions, 2 grams of A must react with 16 grams of B. Undoubtedly, Dalton’s greatest contribution to science was the first scientific theory in history on the composition of matter.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
